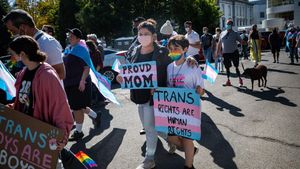Treatment GuideJust DiagnosedSex & DatingAfrican AmericanStigmaAsk the HIV DocPrEP En EspañolNewsVoicesPrint IssueVideoOut 100
CONTACTCAREER OPPORTUNITIESADVERTISE WITH USPRIVACY POLICYPRIVACY PREFERENCESTERMS OF USELEGAL NOTICE
© 2026 Pride Publishing Inc.
All Rights reserved
All Rights reserved
By continuing to use our site, you agree to our Privacy Policy and Terms of Use.
The National Institutes of Health launched a Phase II clinical trial in October of HVTN 205, an experimental preventive HIV vaccine that uses synthetic genes from strains A, B, and C of the virus, which account for 85% of the world's HIV cases. __________ Merck announced plans in September to double the enrollment in its clinical trial of MRKAd5--a preventive and therapeutic experimental HIV vaccine--after early study data showed that the vaccine boosted participants' CD4 cells to 50 to 100 times their baseline number. The vaccine uses a common cold virus to deliver key HIV genes into the body to stimulate the immune system to attack HIV.
From our Sponsors
Most Popular
“So much life to live”: Eric Nieves on thriving with HIV
September 03 2025 11:37 AM
The Talk: Beyond the exam room
August 13 2025 3:15 PM
Messenger RNA could be the key to an HIV vaccine — but government cuts pose a threat
August 20 2025 8:02 AM
It’s National PrEP Day! Learn the latest about HIV prevention
October 10 2025 9:00 AM
Amazing People of 2025: Javier Muñoz
October 17 2025 7:35 PM
The lab coat just got queer
August 21 2025 10:00 AM
The Talk: Owning your voice
August 25 2025 8:16 PM
“I am the steward of my ship”: John Gibson rewrites his HIV narrative
September 16 2025 2:56 PM
Plus: Featured Video
Latest Stories
HIV-positive men stage 'Kiss-In' protest at U.S.-Mexico border
December 01 2025 12:56 PM
What the AIDS crisis stole from Black gay men
December 01 2025 6:00 AM
The Talk: Navigating your treatment
August 01 2025 6:02 PM
The Talk: Starting the conversation
July 25 2025 4:47 PM
Thanks to U=U, HIV-positive people can live long, happy, healthy lives
July 25 2025 2:37 PM
How the Black AIDS Institute continues to fill in the gaps
July 25 2025 1:06 PM
“I felt like a butterfly”: Niko Flowers on reclaiming life with HIV
July 23 2025 12:22 PM
Dancer. Healer. Survivor. DéShaun Armbrister is all of the above
July 02 2025 8:23 PM
BREAKING: Supreme Court rules to save free access to preventive care, including PrEP
June 27 2025 10:32 AM
1985: the year the AIDS crisis finally broke through the silence
June 26 2025 11:24 AM
VIDEO: A man living with HIV discusses his journey to fatherhood
June 10 2025 4:58 PM
Trump admin guts $258 million in funding for HIV vaccine research
June 03 2025 3:47 PM
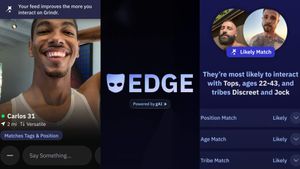
Grindr is reminding us why jockstraps are so sexy and iconic
May 02 2025 5:36 PM
HRC holds 'die-in' to protest Trump health care cuts
April 28 2025 2:11 PM
Two right-wing Supreme Court justices signal they may uphold access to PrEP and more
April 21 2025 4:10 PM
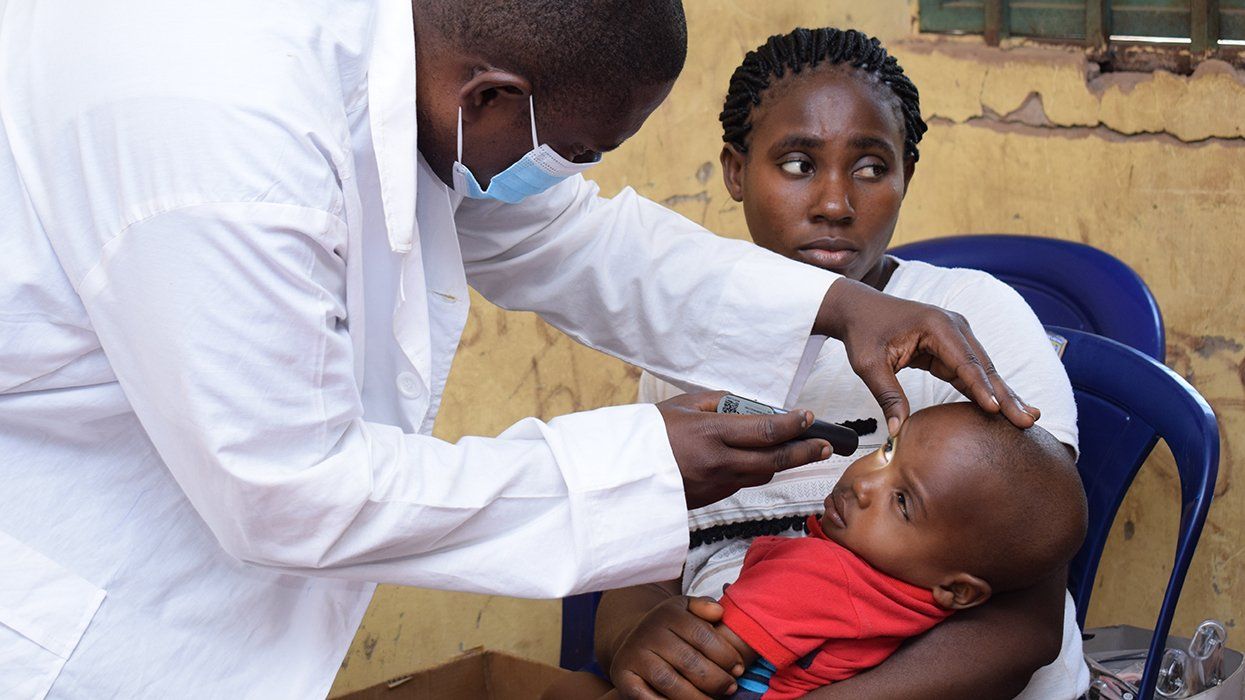
500,000 Children at Risk: PEPFAR Funding Crisis
April 08 2025 3:51 PM
Trending stories
Recommended Stories for You