Treatment GuideJust DiagnosedSex & DatingAfrican AmericanStigmaAsk the HIV DocPrEP En EspañolNewsVoicesPrint IssueVideoOut 100
CONTACTCAREER OPPORTUNITIESADVERTISE WITH USPRIVACY POLICYPRIVACY PREFERENCESTERMS OF USELEGAL NOTICE
© 2025 Pride Publishing Inc.
All Rights reserved
All Rights reserved
By continuing to use our site, you agree to our Privacy Policy and Terms of Use.
The Food and Drug Administration has granted expanded approval to protease inhibitor Prezista for use in first-line therapy among treatment-naive HIVers as well as full approval for its use among treatment-experienced adults. Prezista previously was approved under an accelerated review program only for HIVers with drug resistance. --------------------- A study in the journal AIDS recommends several guidelines to monitor HIVers for emerging resistance to Prezista, including the development of two viral mutations conveying drug resistance and high viral loads after 24 weeks of treatment. Patients who've developed resistance to Prezista also should be monitored for possible cross-resistance to Aptivus, according to the study. --------------------- An analysis in the journal AIDS has shown that newer once-daily, fixed-dose antiretroviral regimens like Epzicom, Atripla, and Truvada are more durable than earlier antiretroviral drugs. Durability is vital to long-term treatment success since anti-HIV medications must be taken indefinitely. --------------------- Data from two long-term studies reported in the journal AIDS confirm the results of previous research that has shown that Viread can negatively affect kidney function, but they also show that kidney toxicity is generally mild and is not linked with the development of more serious renal disease. --------------------- The FDA has announced that it is investigating possible hemarthrosis, a side effect marked by bleeding into the body's joints, with the nonnuke Intelence. The agency also is investigating the hepatitis B treatment Tyzeka for possible peripheral neuropathy. --------------------- Myriad Genetics is launching Phase II human trials of experimental maturation inhibitor vivecon (MPC-9055). The drug, part of an entirely new class of antiretrovirals, targets one of the final stages of viral replication by interfering with the assembly of newly created HIV particles. --------------------- The FDA has made changes to the label of protease inhibitor Norvir to indicate that the drug might cause heart rhythm abnormalities. The label now says Norvir should be used with caution in patients with heart disease or preexisting cardiac abnormalities. --------------------- Merck has launched a study of integrase inhibitor Isentress for use in first-line therapy among treatment-naive HIVers. Isentress is currently approved only for patients with drug-resistant infections. --------------------- Studies of Tibotec experimental nonnuke rilpivirine (TMC278) have shown that the drug might offer months of sustained anti-HIV activity after a single injection of a 'depot formulation' that releases medication in the body slowly over time. Additional studies have been planned. --------------------- A study of the antiepileptic drug Lyrica (pregabalin) for the treatment of HIV-associated peripheral neuropathy has shown that the medication was only slightly more effective in reducing pain scores than a placebo. The medication has been shown to help with other neuropathic pain syndromes. --------------------- HIVers treated with interferon for hepatitis C coinfection could face higher risks for neuropsychiatric side effects if also taking Sustiva, researchers report in the Journal of Acquired Immune Deficiency Syndromes. Combining the two medications boosted reported mood disorders by 50%. --------------------- An analysis of Spanish HIVers has shown that risks for progressive multifocal leukoencephalopathy, a potentially severe brain infection, remain high even in the presence of successful antiretroviral treatment. Many of the 61 PML cases diagnosed occurred among HIVers with undetectable viral loads and CD4-cell counts above 200.
From our Sponsors
Most Popular
“So much life to live”: Eric Nieves on thriving with HIV
September 03 2025 11:37 AM
Thanks to U=U, HIV-positive people can live long, happy, healthy lives
July 25 2025 2:37 PM
The Talk: Beyond the exam room
August 13 2025 3:15 PM
BREAKING: Supreme Court rules to save free access to preventive care, including PrEP
June 27 2025 10:32 AM
Messenger RNA could be the key to an HIV vaccine — but government cuts pose a threat
August 20 2025 8:02 AM
“I felt like a butterfly”: Niko Flowers on reclaiming life with HIV
July 23 2025 12:22 PM
Dancer. Healer. Survivor. DéShaun Armbrister is all of the above
July 02 2025 8:23 PM
The Talk: Starting the conversation
July 25 2025 4:47 PM
The lab coat just got queer
August 21 2025 10:00 AM
Plus: Featured Video
Latest Stories
HIV-positive men stage 'Kiss-In' protest at U.S.-Mexico border
December 01 2025 12:56 PM
What the AIDS crisis stole from Black gay men
December 01 2025 6:00 AM
Amazing People of 2025: Javier Muñoz
October 17 2025 7:35 PM
It’s National PrEP Day! Learn the latest about HIV prevention
October 10 2025 9:00 AM
“I am the steward of my ship”: John Gibson rewrites his HIV narrative
September 16 2025 2:56 PM
The Talk: Owning your voice
August 25 2025 8:16 PM
The Talk: Navigating your treatment
August 01 2025 6:02 PM
How the Black AIDS Institute continues to fill in the gaps
July 25 2025 1:06 PM
1985: the year the AIDS crisis finally broke through the silence
June 26 2025 11:24 AM
VIDEO: A man living with HIV discusses his journey to fatherhood
June 10 2025 4:58 PM
Trump admin guts $258 million in funding for HIV vaccine research
June 03 2025 3:47 PM
Grindr is reminding us why jockstraps are so sexy and iconic
May 02 2025 5:36 PM
HRC holds 'die-in' to protest Trump health care cuts
April 28 2025 2:11 PM
Two right-wing Supreme Court justices signal they may uphold access to PrEP and more
April 21 2025 4:10 PM
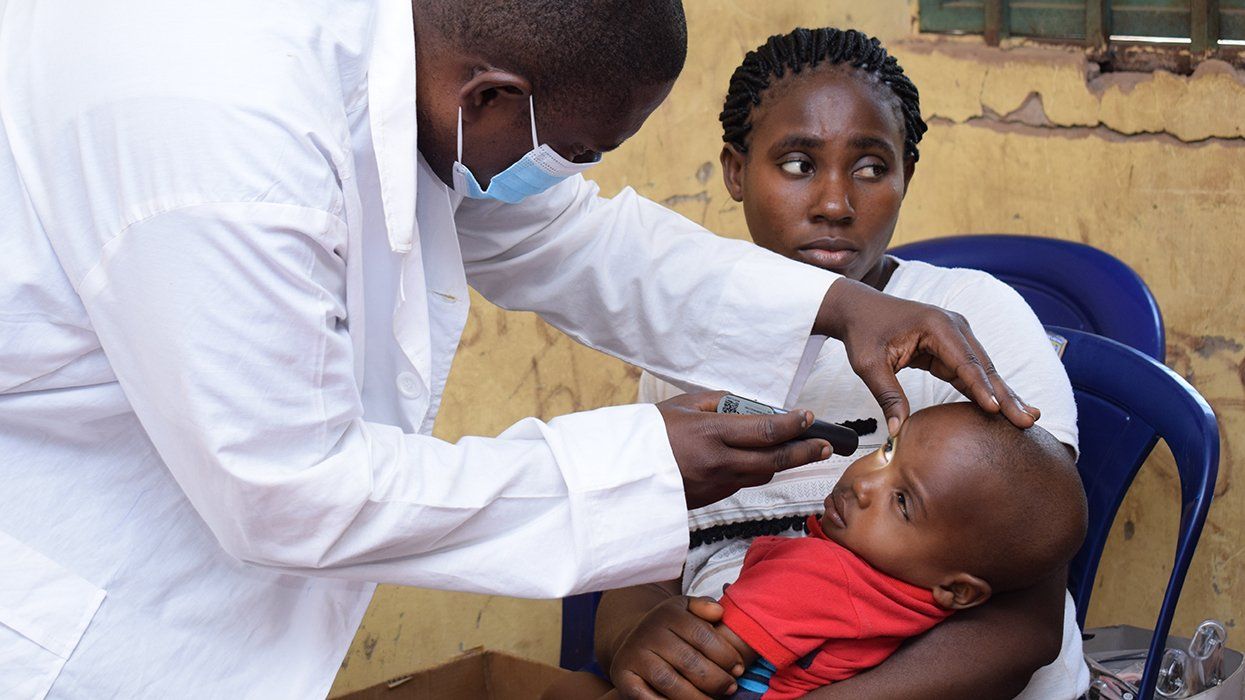
500,000 Children at Risk: PEPFAR Funding Crisis
April 08 2025 3:51 PM
Broadway's best raise over $1 million for LGBTQ+ and HIV causes
April 03 2025 7:15 PM
The Talk Season 5 premieres this spring with HIV guidance for the newly diagnosed
March 26 2025 1:00 PM