September 22 2010 8:46 AM EST
Treatment GuideJust DiagnosedSex & DatingAfrican AmericanStigmaAsk the HIV DocPrEP En EspañolNewsVoicesPrint IssueVideoOut 100
CONTACTCAREER OPPORTUNITIESADVERTISE WITH USPRIVACY POLICYPRIVACY PREFERENCESTERMS OF USELEGAL NOTICE
© 2026 Pride Publishing Inc.
All Rights reserved
All Rights reserved
By continuing to use our site, you agree to our Privacy Policy and Terms of Use.
A new landmark study, which demonstrated it is possible to recruit large numbers of women into a clinical trial evaluating treatments for HIV infection, found no significant gender-based differences in response to the anti-HIV drugs darunavir and ritonavir — at least among those who remained in the trial to the end.
However, women dropped out of the Gender, Race and Clinical Experience study at higher rates than men for reasons other than drug failure, indicating that more must be done to retain women in clinical trials that shed important light on the effectiveness of drugs in development or those on the market.
The study, published in the September 21 issue of Annals of Internal Medicine, showed that nearly one third of women dropped out, compared with less than one-fourth of the men enrolled.
"It is critical that women are involved in clinical trials to evaluate the efficacy and toxicity of new treatment — not just for HIV but for all diseases," says Judith Currier, the study's lead author, who is a professor of medicine and chief of the division of infectious diseases at the David Geffen School of Medicine at UCLA. Currier is also codirector of the UCLA Center for Clinical AIDS Research and Education and a member of the UCLA AIDS Institute.
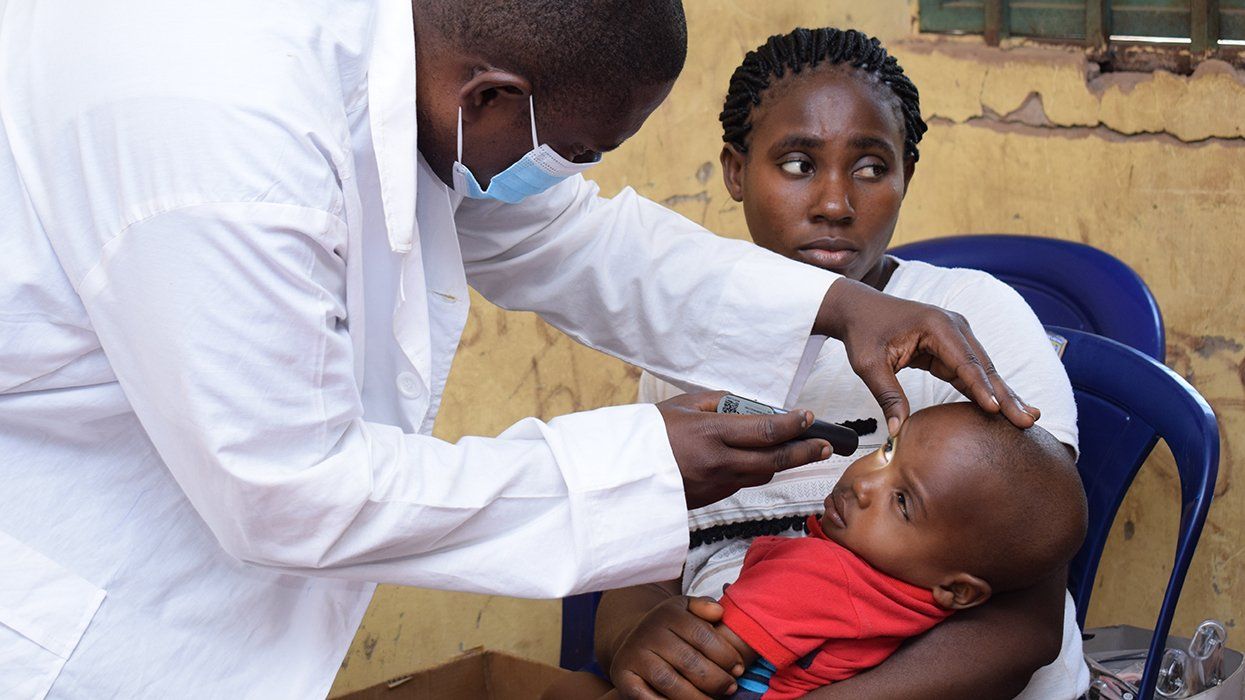
Conducted between October 6, 2006, and December 19, 2008, at 65 sites in the United States, Puerto Rico, and Canada, the study examined the drugs' effectiveness, their adverse effects, and tolerability in both men and women. The researchers recruited 429 HIV-positive subjects who had previously taken HIV therapy. Of the subjects, 287 (66.9%) were women and 142 (33.1%) were men. Overall, 84% were black or Hispanic. In most treatment studies of HIV medications, women account for less than 25% of participants.
The study participants received 600 milligrams of darunavir and 100 milligrams of ritonavir twice daily. In addition, etravirine, tenofovir, and emtricitabine were available for use as needed. Tibotec Therapeutics markets darunavir and etravirine, Abbot Laboratories markets ritonavir, and Gilead markets tenofovir and emtricitabine.
The percentages of women and men who were able to suppress the HIV virus during the study were generally similar. However, a significantly higher proportion of women than men discontinued the study (32.8% versus 23.2%).
When the researchers examined the treatment response of all the participants who entered the trial — including those who eventually dropped out — they found that response rates in women appeared lower than response rates in men (50.9% versus 58.5%). But this difference appeared to be due to the higher dropout rate among women. When they excluded participants who discontinued the study for reasons other than virologic failure, the response rates were similar in women and men (73% versus 73.5%).
Adverse side effects were also comparable between the sexes. For both women and men, the most common side effects — which were considered to be at least possibly related to the treatment — included nausea (5.2% of women, 2.8% of men), diarrhea (4.5% versus 4.9%) and rash (2.1% versus 2.8%).
"The GRACE study shows that it is possible to enroll large numbers of women into HIV treatment trials," the researchers conclude. "The higher discontinuation rate in women, which was driven by reasons other than virologic failure, highlights the need for additional efforts to retain diverse populations in studies."
From our Sponsors
Most Popular
“So much life to live”: Eric Nieves on thriving with HIV
September 03 2025 11:37 AM
The Talk: Beyond the exam room
August 13 2025 3:15 PM
Messenger RNA could be the key to an HIV vaccine — but government cuts pose a threat
August 20 2025 8:02 AM
It’s National PrEP Day! Learn the latest about HIV prevention
October 10 2025 9:00 AM
Amazing People of 2025: Javier Muñoz
October 17 2025 7:35 PM
The lab coat just got queer
August 21 2025 10:00 AM
The Talk: Owning your voice
August 25 2025 8:16 PM
“I am the steward of my ship”: John Gibson rewrites his HIV narrative
September 16 2025 2:56 PM
Plus: Featured Video
Latest Stories
HIV-positive men stage 'Kiss-In' protest at U.S.-Mexico border
December 01 2025 12:56 PM
What the AIDS crisis stole from Black gay men
December 01 2025 6:00 AM
The Talk: Navigating your treatment
August 01 2025 6:02 PM
The Talk: Starting the conversation
July 25 2025 4:47 PM
Thanks to U=U, HIV-positive people can live long, happy, healthy lives
July 25 2025 2:37 PM
How the Black AIDS Institute continues to fill in the gaps
July 25 2025 1:06 PM
“I felt like a butterfly”: Niko Flowers on reclaiming life with HIV
July 23 2025 12:22 PM
Dancer. Healer. Survivor. DéShaun Armbrister is all of the above
July 02 2025 8:23 PM
BREAKING: Supreme Court rules to save free access to preventive care, including PrEP
June 27 2025 10:32 AM
1985: the year the AIDS crisis finally broke through the silence
June 26 2025 11:24 AM
VIDEO: A man living with HIV discusses his journey to fatherhood
June 10 2025 4:58 PM
Trump admin guts $258 million in funding for HIV vaccine research
June 03 2025 3:47 PM
Grindr is reminding us why jockstraps are so sexy and iconic
May 02 2025 5:36 PM
HRC holds 'die-in' to protest Trump health care cuts
April 28 2025 2:11 PM
Two right-wing Supreme Court justices signal they may uphold access to PrEP and more
April 21 2025 4:10 PM
500,000 Children at Risk: PEPFAR Funding Crisis
April 08 2025 3:51 PM